-
Lithium-ion batteries – electrolytes – solid & semi-solid
-
A composite polymer electrolyte was prepared by polymerizing single-ion conductive
monomers with crosslinking agents, followed by EC (ethylene carbonate) infusion. The
electrolyte contains a single-ion conductive polymer polymerized from
LiSTFSI (lithium styrenesulfonyl(trifluoromethanesulfonyl)imide) monomer and
PEGDA (polyethylene glycol diacrylate) crosslinking agent in the presence of
Li6.75La3Zr1.75Ta0.25O12
(LLZTO) inorganic particles.
The single-ion conductive monomer was dissolved in acetonitrile solvent
(10 mass%), followed by addition of LLZTO particles (average particle size:
1 μm, magnetic stirring, room temperature, 18 h). The crosslinking agent
PEGDA (Mn = 575 g/mol) was then added (additional stirring,
room temperature, 10 min). The mixed solution was coated onto a substrate and
heated (60°C, vacuum oven, 18 h) for crosslinking polymerization and solvent
removal to form a film.
The cyclic carbonate EC was liquefied by brief
heating (35-40°C oven) and impregnated into the polymer film. The final
composite electrolyte exhibits a weight ratio of single-ion conductive monomer :
crosslinking agent : inorganic particles : EC =
10 : 3 : 38 : 49.
The composite polymer electrolyte exhibits an ionic conductivity of
8.3 × 10-4 S/cm (25°C). This ionic conductivity was
attributed to the optimized ratio of EC to lithium ions (≥5),
which arguably enhances lithium ion mobility while maintaining mechanical integrity.
It is argued that the electrolyte demonstrates favorable compatibility with lithium metal
negative electrodes, suppressing dendrite formation and improving cycle life
in all-solid-state battery applications (no quantitative data identified).
Although quantitative performance data was not identified aside from ionic conductivity,
this work illustrates how LG Energy Solution evaluates semi-solid composite electrolytes
that exhibit the benefits of solid electrolytes when assembled at room temperature (below
melting point of EC, ≥34°C), i.e. there is no need for a liquid electrolyte filling step
and bi- or multilayer electrolyte architectures are feasible (selection of catholytes
and anolytes that exhibit synergetic properties with respect to positive
and negative electrode active materials).
Although EC might melt during operation (such as during fast charge), it's boiling point
of 248°C is much higher than for example EMC (ethyl methyl carbonate, 107°C), enabling
improved inherent safety characteristics as compared to typical liquid carbonate
electrolyte-based Li-ion battery cells.
An open question is if EC might exhibit elevated SEI formation compared to alternative liquid components,
such as dinitriles (modification of the liquid component is possible even at a relatively late stage
of cell development, compared to other components, given that the infusion step occurs after the
film formation step).
-
The premium version includes another two patent discussions, plus an Excel list with 50-100 commercially relevant recent patent families.
-
Lithium-ion batteries – negative electrode (excluding Li metal electrodes)
-
Silicon nanoparticles (D50: 115 nm, 53 mass%) were prepared through mechanical
milling of silicon raw material (12 h). Flaky graphite with a central particle
size of 8 μm and carbon nanotubes were added at 18 mass% and 0.50 mass%,
respectively. The carbon nanotubes exhibited a BET specific surface area of
645 m2/g and a bulk density of 0.057 g/cm3.
The mixture was combined with petroleum pitch (fixed carbon ratio: 77.7%,
beta resin value: 28.1%) and dispersed using a high-speed mixer. The blend was
then subjected to uniaxial compression molding at 0.64 ton/cm2
pressure to form compacts.
The compacts were carbonized at 950°C for 4 h under an inert atmosphere,
followed by jet milling to achieve a D50 particle size of 10-15 μm. The
resulting silicon-carbon composite exhibits a minimum pore size of
>10 nm and ≤30 nm as determined by DFT pore analysis.
A surface carbon coating was applied by mixing the composite with coal tar
using a twisted blade mixer (30 min), followed by heat treatment at 1,000°C
under inert atmosphere. The final material was sieved through a 20 μm mesh.
For electrochemical evaluation, negative electrodes were prepared by mixing the
silicon-carbon composite with commercial spherical natural graphite
(15 : 85 mass ratio) to achieve a target capacity of 550 mAh/g. The
electrode composition was active material : conductive additive : CMC : SBR =
96.1 : 1 : 1.7 : 1.2 mass ratio, with a loading of ≈7 mg/cm2 and
pressed density of 1.35 g/cm3.
The optimized material exhibits a BET specific surface area of
4.03 m2/g, a discharge capacity of 554 mAh/g, a first cycle
efficiency of 90.2%, and a capacity retention of 87.1% after 50 cycles
(0.5 C charge / discharge).
This work illustrates an evolution in the materials employed by Posco
for Si-carbon composite formation while retaining the uniaxial compression
molding process step in this product development track,
along with metallurgical Si as starting material.
-
The premium version includes another two patent discussions, plus an Excel list with 50-100 commercially relevant recent patent families.
-
Lithium-ion batteries – positive electrode
-
A positive electrode active material was prepared using
LiMn0.6Fe0.4PO4 as the base olivine structure,
doped with titanium, vanadium, and niobium to improve particle size control
and rolling (pressed) density. The material was synthesized through a controlled
calcination process involving metal precursor compounds.
Commercial lithium manganese iron phosphate (LiMn0.6Fe0.4PO4)
was mixed with titanium dioxide (TiO2), ammonium metavanadate
(NH4VO3), and niobium oxide (Nb2O5) in
specific molar ratios (0.01 mol% Ti, 0.01 mol% V, and 0.01 mol% Nb relative to the total
metal content excluding lithium).
Prior to mixing with metal precursors, the base LMFP material underwent
pre-calcination (700°C, 1 h, nitrogen atmosphere) to remove moisture and
improve crystallinity. The pre-treated material was then combined with the
metal precursor compounds and calcined again under the same conditions.
The crystallite size was measured at 89 nm, which is claimed as an optimal
grain size for mechanical properties. Particle size distribution analysis
exhibits a D50 of 0.85 μm and a D90/D10 ratio of
7.2.
Rolling density measurements at 9,000 kgf pressure exhibit
2.41 g/cc, significantly higher than undoped LMFP (2.28 g/cc).
In half-cells, the material exhibits an initial discharge capacity of
151.7 mAh/g with a first cycle efficiency of 97.4%, along with a capacity retention
after 50 cycles of 97.4% (0.5 C charge/discharge).
This work illustrates that LG Energy Solution pursues a multi-element Ti / V / Nb doping approach
to achieve well-balanced electrochemical performance with LMFP active materials.
-
The premium version includes another two patent discussions, plus an Excel list with 50-100 commercially relevant recent patent families.
-
Fuel cells (PEMFC / SOFC / PAFC / AEMFC) – electrochemically active materials
-
A carbon black support was processed using a multi-step
hybrid activation method to produce high surface area catalyst supports for
fuel cells.
The high-temperature, high-pressure rotary kiln-type reactor
was tilted at 10-30° with the steam and CO2 inlet
positioned at the bottom to enhance gas circulation. Carbon black
(BET specific surface area: 130 m2/g, average particle size: 23 nm)
was introduced into the vacuum-evacuated reactor via vacuum suction.
The reactor was sealed and nitrogen was injected to remove oxygen. The
carbon black support was then activated by injecting steam and
CO2 while heating (1,050°C, 3 h, 0.8 bar pressure). The reactor
was maintained in a saturated state with the calculated amounts of steam and
CO2.
After activation, product gases were removed under reduced pressure while
maintaining the temperature (1,050°C, 30 min). This gas removal step is
critical for enabling deeper pore development in the subsequent reactivation.
The carbon support was then reactivated by re-injecting steam and
CO2 under pressure (1,050°C, 5 h, 0.8 bar). This reactivation
step created fine micropores and increased the surface area significantly.
The resulting catalyst support exhibits a
BET specific surface area of
755 m2/g. A comparative example using extended
activation without gas removal exhibits only 628 m2/g,
demonstrating the critical importance of the gas removal step. TEM analysis
confirms retention of high crystallinity with crystallinity (Lc) of 2.46 nm.
A platinum catalyst (40 mass% loading) was subsequently deposited on this
support using platinum chloride and monoethanolamine at 160°C in ethylene glycol (processing using
homogenizer and ultrasonic treatment to achieve particle sizes below 30 nm).
Fuel cell performance testing reveals superior unit cell performance compared
to catalysts prepared on conventional carbon supports. In single cell tests
(65°C, 100% relative humidity, 25 cm2 active area), the optimized
catalyst exhibits enhanced current density across the entire voltage
range as shown in the Figure below (blue: example, gray: comparative example).
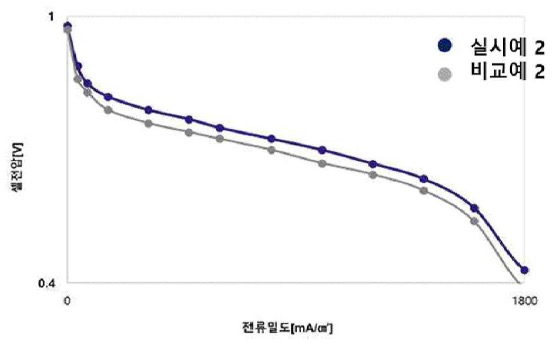
This work illustrates how a 2-step activation process with intermediate gas removal leads to carbon support materials
with improved pore structure and BET surface area, resulting in enhanced PEMFC performance.
-
The premium version includes another two patent discussions, plus an Excel list with 50-100 commercially relevant recent patent families.
-
Triweekly patent lists for other categories (Excel files are included for premium users)
-
- Lithium metal batteries (excluding Li-S, Li-Air): XLSX
-
- Lithium-air batteries: XLSX
-
- Lithium-ion batteries – electrolytes – liquid: XLSX
-
- Lithium-ion batteries – separators: XLSX
-
- Lithium-sulfur batteries: XLSX
-
- Na-ion batteries: XLSX
-
Prior patent updates
-
2025-07-22
-
2025-07-01
-
2025-06-10
-
2025-05-20
-
2025-04-29
|